Difference Between Atomic and Molecular Spectroscopy
Science has continuously changed the way we think about things and has made most, if not all things in the universe related to us much more clearer and easier to understand. Many years ago, it was not easy to understand what things are made up of and how molecules and atoms form everything around us, including our bodies and basically all that we can see and touch. For many people, these concepts are still very mind boggling, but that may be because they are ignorant when it comes to actually studying them.
Spectroscopy is basically the study of the interaction between matter and radiated energy. This concept originated during the study of visible light and other ideas that have now evolved into much more conclusive facts and theories. There are a number of materials that spectroscopic studies deal with, like atoms, molecules, crystals and nuclei. While they are not entirely limited to these materials, they are definitely dependent on them to some extent.
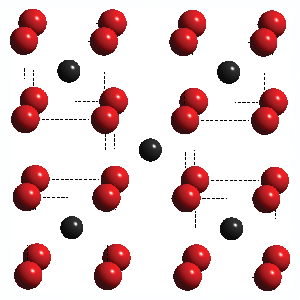
The first application of spectroscopy that was developed was atomic spectroscopy. This type of spectroscopy deals with atomic absorption, atomic emission and various things such as atomic spectral lines and electronic transitions. This is the major difference between atomic and molecular spectroscopy as the former is only concerned with the properties of atoms, while the latter deals with molecules, which are infinitely more in number.
Finding the nature of atoms is something that is done with atomic spectroscopy while molecular spectroscopy deals with the interaction of electromagnetic waves, chemical functions and structure of the matter in question. The two may be similar to some extent, but are usually classified to be very different.
When dealing with the nature of interactions, the two types that are being discussed are crucial to the study and in determining whether the form of the matter is true in nature or not.
Instructions
-
1
Atomic Spectroscopy
The field of spectroscopy which deals with atoms and looking at the energy levels of electrons around a single atom or sometimes small molecules is termed atomic spectroscopy.
Image Courtesy: nist.gov
-
2
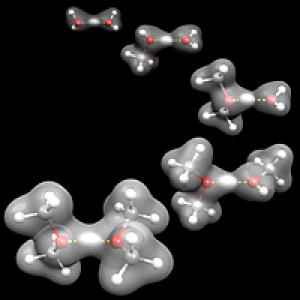
Molecular Spectroscopy
When talking about bond lengths and vibrational modes of matter and the interaction of electromagnetic waves with the matter in hand, you are dealing with molecular spectroscopy, which is much more complicated.
Image Courtesy: lanl.gov